 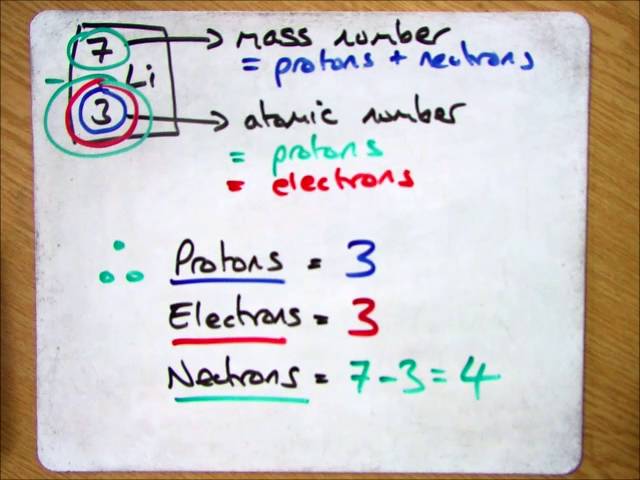
As a result, as the number of protons increases, an increasing ratio of neutrons to protons is needed to form a stable nucleus. Neutrons stabilize the nucleus, because they attract each other and protons, which helps offset the electrical repulsion between protons. There are only certain combinations of neutrons and protons, which forms stable nuclei. These two forces compete, leading to various stability of nuclei. The neutron has a mean square radius of about 0.8×10−15 m, or 0.8 fm, and it is a spin-½ fermion.Ītomic nuclei consist of protons and neutrons, which attract each other through the nuclear force, while protons repel each other via the electric force due to their positive charge. It has no electric charge and a rest mass equal to 1.67493 × 10−27 kg-marginally greater than that of the proton but nearly 1839 times greater than that of the electron. In the universe, neutrons are abundant, making up more than half of all visible matter. Moscovium does not occur in natural isotope.Ī neutron is one of the subatomic particles that make up matter. Mass numbers of typical isotopes of Moscovium are 287-290. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. 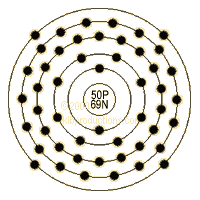 
Moscovium is a chemical element with atomic number 115 which means there are 115 protons in its nucleus. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
